Radiometric dating can take a number of different forms. In most cases, it involves measuring certain radioactive isotopes and their daughter products and a fairly simple half-life calculation which tells us how long it would normally take, at current rates of decay, to produce the known amount of daughter product. This part is all quite straightforward, but there are some assumptions that go into radiometric dating.
One of the major assumptions is that the rate of radioactive decay has always been the same. It’s an understandable assumption because the rate of decay is primarily a function of physics and it is reasonable to assume that physical laws do not vary.
However, we do have evidence from conflicting “clocks” that while extensive amounts of radioactive decay have occurred, they did not take the usual amount of time we would expect from today’s rates. This evidence indicates that the rate of decay was much faster at some point in the past.
For example, we know that during the formation of zircon crystals, the crystal lattice excludes lead, but can incorporate uranium. Zircon crystals form a good system for radiometric dating because uranium decays to lead and we have good evidence that lead would not be found in zircon crystals at their formation. Thus, any lead in a zircon crystal would have come from the decay of uranium to lead rather than being present from the start.
In addition to this, the decay of uranium to lead causes alpha particles to be given off by the decaying nucleus. Alpha particles are composed of 2 protons and 2 neutrons and are the same thing as the nucleus of a helium atom. This is important in two ways.
First, the ejected particle causes discernible damage to the crystal structure which can be seen as fission tracks. These help verify that the lead present in the crystal was formed from the decay of uranium because if it had been there already, during the crystal formation, there would be no fission tracks.
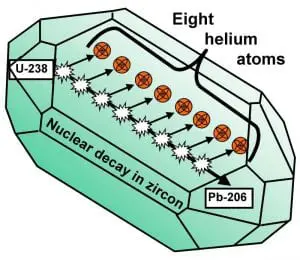
Photo used with permission from Dr. Russ Humphreys
Second, the alpha particles pick up free electrons to become normal helium atoms. Helium is inert and thus does not react with other substances. It is also a very small and light atom that diffuses rapidly out of the crystals. At known rates, during normal radioactive decay, the rate of helium diffusion is faster than the rate of production. It should never be able to build up. Think of pouring water into a bucket with a hole in it. If the hole is big, you can pour water in and the bucket will never be full because it pours out the hole as fast as you are pouring it in. Zircon crystals are like a bucket with a big hole in it when it comes to helium because the helium leaves as fast as it is produced at normal rates of production.
However, what we find is that these zircon crystals not only have lots of lead and fission tracks, indicating that a lot of radioactive decay has occurred, but they also have a lot of helium built up inside the crystal. That shouldn’t be able to happen if radioactive decay has always occurred at today’s rates. So we have two conflicting molecular clocks in the same zircon crystals. One clock says a lot of decay occurred because there’s lots of daughter product, which would usually take millions of years to produce. The other clock says the decay must have happened rapidly because in order to have helium built up inside the crystal, the rate of helium production had to be faster than the rate at which helium diffuses out of the crystal. The input had to be faster than the output or it would never build up. Since zircons lose helium quickly, the rate of helium production had to be really fast at some point in the past, and even now that rates are slower, there is still helium left that hasn’t had time to diffuse out yet. Or in other words, the bucket is still half full even though we know the water is pouring out the hole faster than it is coming in right now.
The best explanation for these observations is that the input – the rate of helium production, and thus the rate of radioactive decay – was faster at some point in the past and thus allowed helium to accumulate inside the crystals.
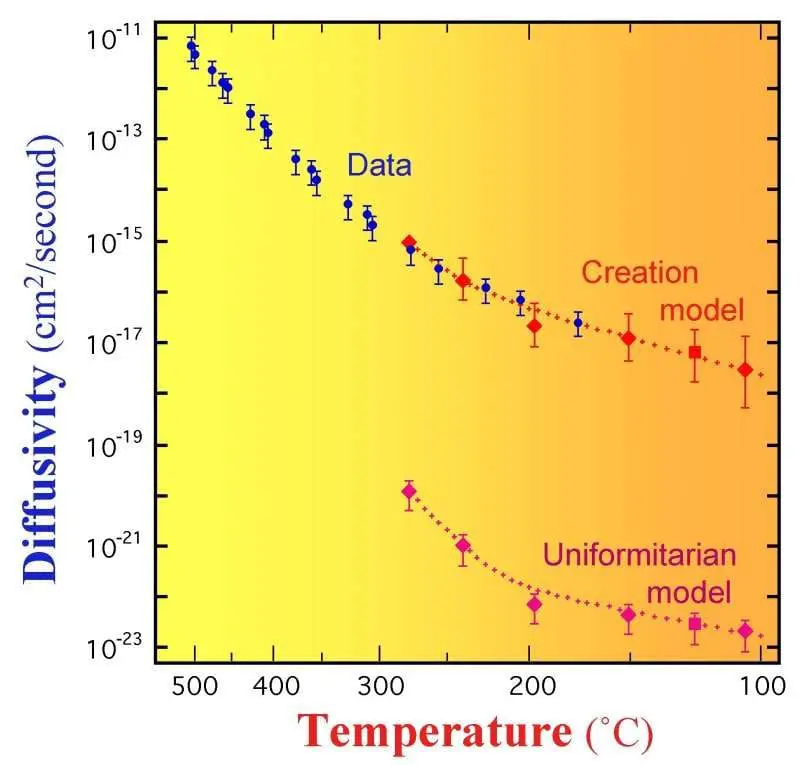
This is not speculation or cherry picking or wishful thinking. It’s an honest attempt to explain ALL of the data. Traditional radiometric dating methods only take into account the amount of daughter product and perhaps the fission tracks, but the helium content has been largely ignored or dismissed. When we take into account the entirety of the data, it is best explained by a period of accelerated radioactive decay at some point in the past. What’s more, the result of taking into consideration all of the data, including helium diffusion rates, produces an age for these mineral crystals that is very consistent with the predictions of a young earth timeline. In fact, while young earth predictions fit the data very well, old earth predictions are off by a factor of 100,000! The preponderance of the evidence shows an earth that is only a few thousand years old.
References:
- Humphreys, D. R. 2002. Nuclear Decay: Evidence For A Young World. Acts & Facts. 31 (10).
- Humphreys, D. R. 2003. New RATE Data Support a Young World. Acts & Facts. 32 (12).
- Humphreys, D. R. 2005. Young Helium Diffusion Age of Zircons Supports Accelerated Decay. Radioisotopes and the Age of the Earth: Volume II. Larry Vardiman, Andrew A. Snelling, Eugene F. Chaffin, eds. Institute for Creation Research. 25-100.







Dr. Humphreys work is rejected not just by secular scientists, but also a diverse group of young- and old-Earth creationists, including members of the American Scientific Affiliation (ASA). This is primarily because his work is based on very bad, often easily disprovable assumptions as well as questionable, even potentially fraudulent data.
A comprehensive debunking of his work can be found here:
http://www.talkorigins.org/faqs/helium/zircons.html
GREAT ARTICLE Lindsay!
Now if only we could communicate this information for a seventh grader who heard about C14 for the first time in Science Class this past September.
Could you possibly summarize the main points in a group of 45 second videos – use KIDS in the video to explain back to you what you have shared – and send the videos to Janessa Cooper!
GOOD STUFF!
Francis,
I will candidly admit that many criticisms have been leveled at Dr. Humphreys’ work, but they have been addressed repeatedly. Many of the concerns were actually addressed in the second RATE volume from the beginning, which many critics have apparently not read. Others have been addressed since then. Many of the factors that have been alleged to have been ignored were not ignored, but rather were taken into consideration and found to be insignificant.
It is also worth repeating that the data were collected by an independent lab which specializes in just such work and which was not made aware of the intent of the research in order that the experiment would be blind. The young earth predictions were published several years in advance of the data collection. If the experiment were as flawed and useless as many critics have claimed, then it is phenomenally lucky that the data just happened to match the previously published predictions, on par with winning the lottery on the first try. A better explanation is that the researchers were not lucky; they were right.
If you’re interested in reading some of the responses of Dr. Humphreys to his critics, here is an overview of the main criticisms and Dr. Humphreys’ response, with reference to where these responses have been published, many of which appeared in peer-reviewed publications. And here is another page that gives an overview with links as well.
The secular world tries to beat young earth believers over the head with radiometric evidence, but young earth believers need to recognize their weapon is no more than a wet noodle. In actuality, young earth believers should be storming the soap boxes to proclaim the truth. The evidence is on our side – from the many bad assumptions required by the process, to the contradictory evidence provided by other dating methods such as the Helium leak rate as you point out, to the inconsistencies between the various radiometric dating methods, there is no reason to cower before the “radiometric dating proves an old earth” charge. As in all other matters of the faith, we should boldly and proudly be proclaiming the truth.